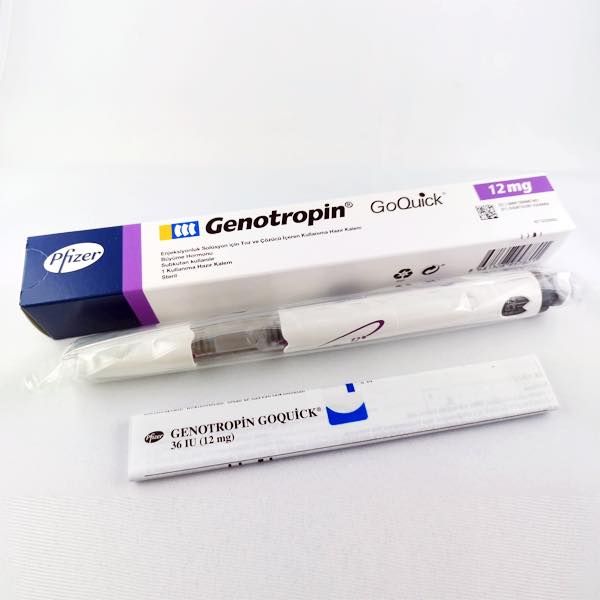
Genotropin 36iu cartridges
$110.00 Original price was: $110.00.$105.00Current price is: $105.00.
BRAND: PHZER
QUANTITY: 1 PEN OF 12MG
Genotropin 36 IU is a premium, ready-to-use cartridge of recombinant human growth hormone (somatropin) designed for use with the Genotropin GoQuick® pen. It is prescribed for the treatment of growth hormone deficiency in children and adults, and for specific medical conditions. This is a controlled, prescription-only medication requiring diagnosis and monitoring by an endocrinologist.
A Benchmark in Growth Hormone Replacement Therapy
Genotropin 36 IU represents a state-of-the-art, FDA-approved pharmaceutical formulation of recombinant human growth hormone (somatropin). Supplied in a convenient, pre-filled, two-chamber cartridge, it is specifically designed for use with the accompanying Genotropin GoQuick® injection pen. This system ensures accurate dosing, sterility, and ease of administration for patients requiring long-term growth hormone therapy to correct a documented hormonal deficiency.
Primary Medical Indications:
-
Pediatric Growth Hormone Deficiency (GHD): Treatment of children who fail to grow due to inadequate endogenous growth hormone secretion.
-
Adult Growth Hormone Deficiency (AGHD): Replacement therapy in adults with confirmed GHD, which can improve body composition, bone density, lipid metabolism, and quality of life.
-
Other Approved Conditions: Including Turner syndrome, Prader-Willi syndrome, chronic renal insufficiency pre-transplant, and children born small for gestational age (SGA) who have not caught up in growth by age 2-4.
Key Features of the Genotropin GoQuick® System:
-
Pre-filled, Dual-Chamber Cartridge: Contains lyophilized somatropin powder in one chamber and a diluent in the other. The simple “click-mix” process in the pen reconstitutes the hormone immediately before use, ensuring maximum potency and stability.
-
GoQuick® Pen: A discreet, easy-to-use, dial-a-dose injection pen. It minimizes preparation steps, reduces dosing errors, and features a hidden needle to reduce anxiety.
-
Dosing Accuracy: Allows for precise, individualized dosing in increments as small as 0.1 mg (which corresponds to specific IU values).
-
Convenience & Compliance: The integrated system greatly simplifies the injection process, improving patient compliance for a therapy that is typically administered daily via subcutaneous injection.
Critical Safety and Prescription Information
Genotropin is a powerful hormonal therapy with significant risks if misused. It is not approved or safe for anti-aging, athletic enhancement, or weight loss.
-
Prescription & Monitoring Mandatory: Treatment must be initiated and strictly supervised by an endocrinologist. Diagnosis requires specific stimulation tests (for GHD). Regular monitoring of blood sugar, thyroid function, IGF-1 levels, and potential side effects is required.
-
Serious Side Effects & Risks: Include increased risk of malignancy progression, intracranial hypertension (severe headache, vision changes), pancreatitis, severe fluid retention, carpal tunnel syndrome, insulin resistance, and worsening of pre-existing diabetes.
-
Contraindications: Active malignancy, critical illness due to complications after open heart or abdominal surgery, multiple trauma, or acute respiratory failure. Not for use in patients with closed epiphyses (growth plates) whose growth is complete, unless for AGHD.
Product Specifications:
-
Active Ingredient: Somatropin (rDNA origin) – 36 IU per cartridge.
-
Form: Dual-chamber cartridge for use with the GoQuick® pen.
-
Administration: Subcutaneous injection.
-
Storage: Unmixed cartridges must be stored refrigerated at 2°C to 8°C (36°F to 46°F). Do not freeze. After mixing in the pen, the product is stable for a limited time as per manufacturer’s instructions (typically 28 days when refrigerated).
-
Prescription Required: YES – A valid prescription from a licensed endocrinologist or specialist is mandatory.
A Commitment to Legitimate Medical Care
Genotropin is a life-changing therapy for individuals with legitimate, diagnosed medical conditions. Our platform supports access only for patients with verified prescriptions and under a doctor’s ongoing care.
The Path to Treatment Begins with a Specialist
Symptoms of growth hormone deficiency are non-specific. Self-diagnosis and treatment are dangerous and illegal. If you suspect a hormonal issue, consult an endocrinologist for a full evaluation. This product information is not medical advice.
For the Web Builder/SEO & Compliance Perspective:
-
Strictly Medical SEO: Target only condition-specific and treatment-specific keywords. Examples: “growth hormone deficiency treatment,” “Genotropin for adults,” “pediatric endocrinology,” “somatropin prescription.” Actively avoid and disavow any keywords related to “anti-aging HGH,” “HGH for muscle building,” “fountain of youth,” or “weight loss HGH.”
-
Build Authority in Endocrinology: Create a robust “Endocrine Health” content section featuring articles written or reviewed by medical professionals. Topics: “Understanding Growth Hormone Deficiency in Adults,” “The Diagnosis Process for GHD,” “Living with Turner Syndrome,” and “Guide to Injection Devices for Hormone Therapy.” This establishes critical E-A-T.
-
Emphasize the Delivery System: The GoQuick® Pen is a major differentiator and compliance aid. Use high-quality images/videos showcasing its ease of use. This addresses a key patient concern (fear of injections) and highlights a genuine product benefit.
-
Highest-Level Prescription Verification: This is a high-cost, high-risk medication. The verification process must be rigorous. It should involve a specialist telemedicine consultation and require the upload of recent diagnostic test results (stimulation tests, IGF-1 levels) and/or a current prescription from a treating endocrinologist.
-
Prominent Safety Warnings: The warnings about malignancy risk, intracranial hypertension, and fluid retention must be in a highlighted, unmissable section. Schema markup must include these as serious
warningproperties. -
Crucial Storage Information: Clearly state the refrigeration requirement before and after mixing. This is a key logistical point for shipping and patient education. Failure here leads to product degradation and patient harm.
-
Structured Data for Medical Devices & Drugs: Use combined
DrugandMedicalDevice(for the pen) schema. For the drug:activeIngredient,dosageForm,prescriptionStatus,warning. For the device:descriptionhighlighting ease of use and accuracy.
Be the first to review “Genotropin 36iu cartridges” Cancel reply
Related products
Human Growth (HGH)
Human Growth (HGH)
Human Growth (HGH)






Reviews
There are no reviews yet.