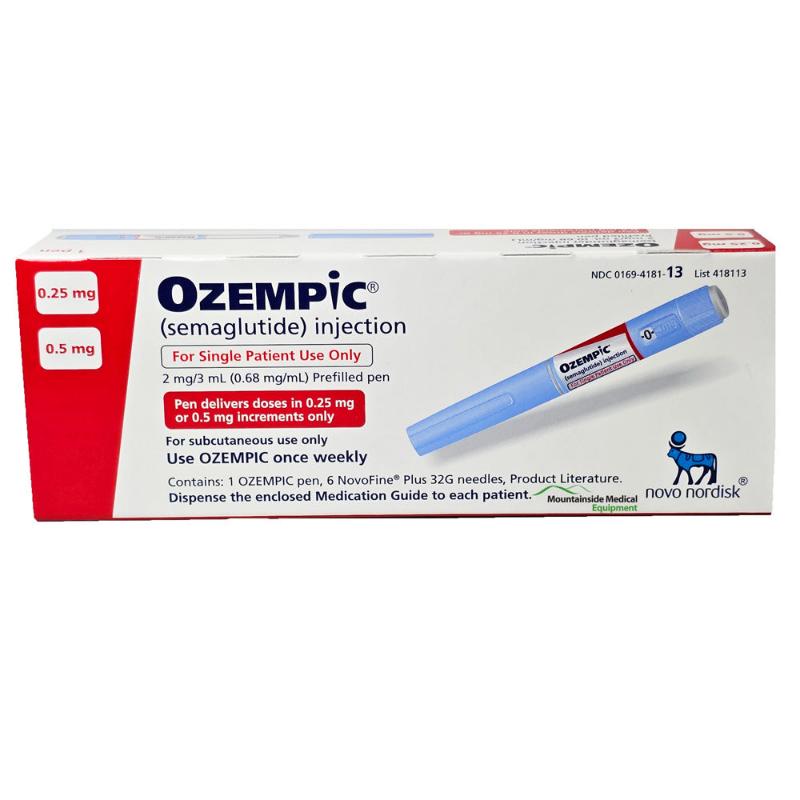
Ozempic 0.25mg
$200.00
The Ozempic® 0.25mg dose is the recommended starting dose for adults beginning treatment for type 2 diabetes. Delivered via a convenient pre-filled pen, this once-weekly initial dose helps your body adjust to semaglutide therapy while starting to improve blood sugar control under medical supervision.
The Foundational Starting Dose for Your Ozempic® Journey
The Ozempic® 0.25mg dose represents the crucial first step in a structured treatment pathway for managing type 2 diabetes. Specifically designed as a four-week initiation dose, this strength allows patients to gradually acclimate to semaglutide therapy—a GLP-1 receptor agonist—while minimizing potential gastrointestinal side effects. Delivered through Novo Nordisk’s proprietary pre-filled pen, this starter dose introduces the body to the medication’s mechanism in a measured, tolerable way, establishing the foundation for effective long-term glycemic management. Healthcare providers prescribe this initial regimen to safely build therapeutic tolerance before escalating to higher maintenance doses.
Gentle Introduction with Early Therapeutic Benefits
The 0.25mg once-weekly dose serves a dual purpose: it initiates the physiological adaptation process while beginning to engage the medication’s primary mechanisms. At this strength, Ozempic® starts to mimic the natural GLP-1 hormone, gently stimulating insulin secretion in response to elevated blood glucose and reducing inappropriate glucagon release from the liver. Patients often notice early effects such as moderated appetite and initial improvements in post-meal blood sugar readings during these first four weeks. This gradual introduction is clinically proven to enhance tolerability, with many patients experiencing fewer or milder gastrointestinal symptoms compared to starting at higher doses, thereby supporting treatment adherence from the very beginning.
Key Features of the 0.25mg Starter Dose:
-
Tolerability-Focused Initiation: Specifically formulated as a starting dose to minimize common side effects like nausea, vomiting, and diarrhea while your body adjusts to therapy.
-
Structured Treatment Pathway: Follows evidence-based clinical guidelines recommending four weeks at 0.25mg before dose escalation, ensuring safe and effective treatment progression.
-
Early Action Mechanism: Begins activating GLP-1 receptors to improve pancreatic insulin response and reduce hepatic glucose production, establishing foundational glycemic control.
-
Same Convenient Delivery: Uses the identical, user-friendly Ozempic® pre-filled pen device as higher doses, allowing patients to become familiar with the injection process from day one.
-
Psychological Preparation: Helps patients establish the routine of once-weekly administration while building confidence in self-injection techniques under medical guidance.
Usage Protocol & Important Safety Information:
-
Dosage Regimen: One subcutaneous injection of 0.25mg once weekly for the first four weeks of treatment. Inject on the same day each week, with or without meals.
-
Administration Sites: May be injected into the abdomen, thigh, or upper arm. Rotate injection sites weekly to reduce the risk of lipodystrophy.
-
Device Features: The Ozempic® 0.25mg dose comes in a pre-filled, multi-dose pen containing sufficient medication for the initial four-week period. The pen features a hidden needle, simple dose selection, and an easy-to-press injection button.
-
Titration Schedule: After four weeks at 0.25mg, the dose should be increased to 0.5mg once weekly under healthcare provider guidance. Do not exceed the 0.25mg dose beyond the initial four weeks unless specifically directed by your physician.
-
Common Side Effects at Initiation: Nausea, diarrhea, vomiting, abdominal pain, and constipation may occur but are typically mild to moderate and diminish over time as the body adjusts.
-
Important Contraindications: Not for patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Not for treatment of type 1 diabetes or diabetic ketoacidosis.
-
Medical Supervision Required: This medication requires a prescription and ongoing monitoring by a healthcare professional. Regular follow-up is essential to assess tolerability, efficacy, and determine appropriate dose escalation.
Be the first to review “Ozempic 0.25mg” Cancel reply
Related products
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes








Reviews
There are no reviews yet.