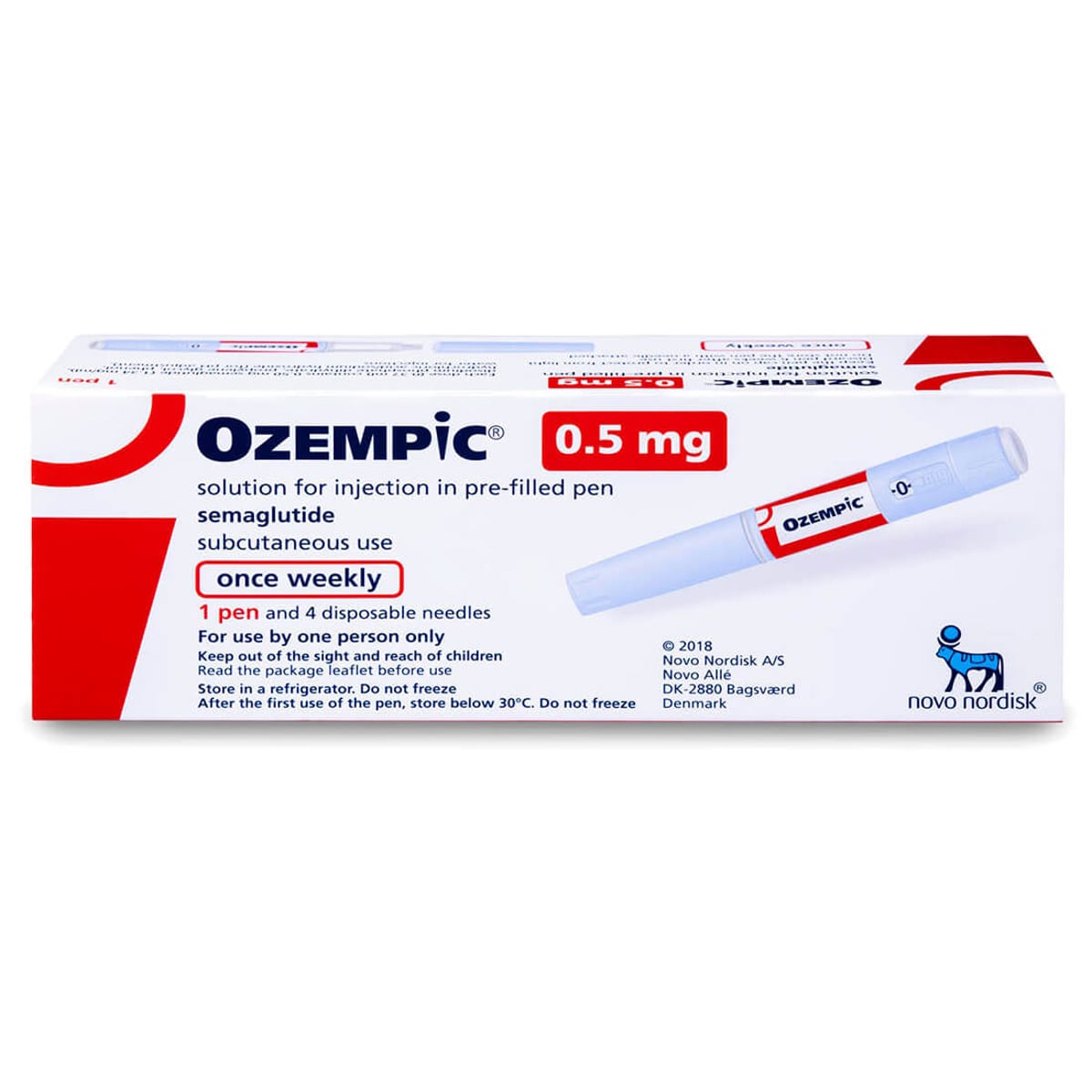
Ozempic 0.5mg
$200.00
The Ozempic® 0.5mg dose is the first therapeutic maintenance dose for adults with type 2 diabetes, following the initial 4-week starter period. This once-weekly subcutaneous injection provides effective blood sugar control and A1C reduction as part of a continued, doctor-supervised treatment plan.
The First Therapeutic Step in Sustained Diabetes Management
The Ozempic® 0.5mg dose marks the transition from treatment initiation to active therapeutic management for type 2 diabetes. Following the recommended four-week period at the 0.25mg starter dose, patients typically advance to this 0.5mg weekly maintenance level under medical guidance. This dosage represents the first potency level at which the full glucose-lowering effects of semaglutide—a GLP-1 receptor agonist—become consistently clinically significant for most patients. Delivered via the same user-friendly pre-filled pen, the 0.5mg dose establishes a foundation for sustained glycemic control, offering a balance of efficacy and tolerability that supports long-term treatment adherence and metabolic health.
Clinically Effective Glycemic Control with Enhanced Benefits
At the 0.5mg weekly strength, Ozempic® delivers meaningful activation of GLP-1 receptors, producing substantial improvements in multiple facets of diabetes management. This dose effectively stimulates glucose-dependent insulin secretion from pancreatic beta cells while suppressing inappropriate glucagon release from the liver—a dual mechanism that addresses both fasting and postprandial hyperglycemia. Many patients experience more pronounced effects on appetite regulation and gastric emptying at this dose, which often translates to continued weight stabilization or reduction. Clinical studies demonstrate that the 0.5mg dose produces statistically significant reductions in hemoglobin A1C, typically in the range of 1.3-1.5% from baseline when combined with diet and exercise, establishing it as a cornerstone of effective type 2 diabetes pharmacotherapy.
Key Features of the 0.5mg Maintenance Dose:
-
Proven Glycemic Efficacy: Delivers clinically meaningful reductions in A1C and fasting plasma glucose levels, helping patients move toward their target glycemic ranges.
-
Weight Management Support: Enhanced GLP-1 receptor activation at this dose frequently contributes to moderate, sustained weight loss—addressing a key comorbidity in type 2 diabetes.
-
Tolerability-Optimized Escalation: Following the 0.25mg initiation period, most patients transition smoothly to 0.5mg with minimal gastrointestinal side effects, supporting continued treatment adherence.
-
Cardiovascular Foundation: Provides the therapeutic level at which cardiovascular outcome benefits begin to manifest in patients with established cardiovascular disease.
-
Flexible Treatment Pathway: Serves as either an effective long-term maintenance dose for many patients or a step toward higher doses (1mg or 2mg) if additional glycemic control is needed.
Usage Protocol & Clinical Considerations:
-
Standard Dosage Regimen: One subcutaneous injection of 0.5mg once weekly, typically following four weeks of treatment at 0.25mg. Administer on the same day each week, with or without meals.
-
Treatment Duration: This dose may be used as a long-term maintenance therapy for many patients. Healthcare providers will assess glycemic response after approximately 4-8 weeks to determine if dose escalation is warranted.
-
Administration: Use the Ozempic® pre-filled pen designed for 0.5mg doses. Inject subcutaneously into the abdomen, thigh, or upper arm, rotating injection sites weekly.
-
Dose Escalation Criteria: If glycemic targets are not achieved after 8 weeks at 0.5mg, the dose may be increased to 1mg once weekly under medical supervision. Do not adjust dosage without consulting your healthcare provider.
-
Common Side Effects: Gastrointestinal symptoms (nausea, diarrhea, vomiting, constipation) may occur but are generally less frequent and severe than during initial therapy. These typically diminish over time.
-
Important Monitoring Parameters: Regular assessment of blood glucose, A1C, renal function, and pancreatic enzymes is recommended. Patients should be monitored for signs of pancreatitis or worsening diabetic retinopathy.
-
Safety Contraindications: Not for patients with personal/family history of medullary thyroid carcinoma or MEN 2. Use with caution in patients with renal impairment or history of pancreatitis.
Be the first to review “Ozempic 0.5mg” Cancel reply
Related products
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes







Reviews
There are no reviews yet.