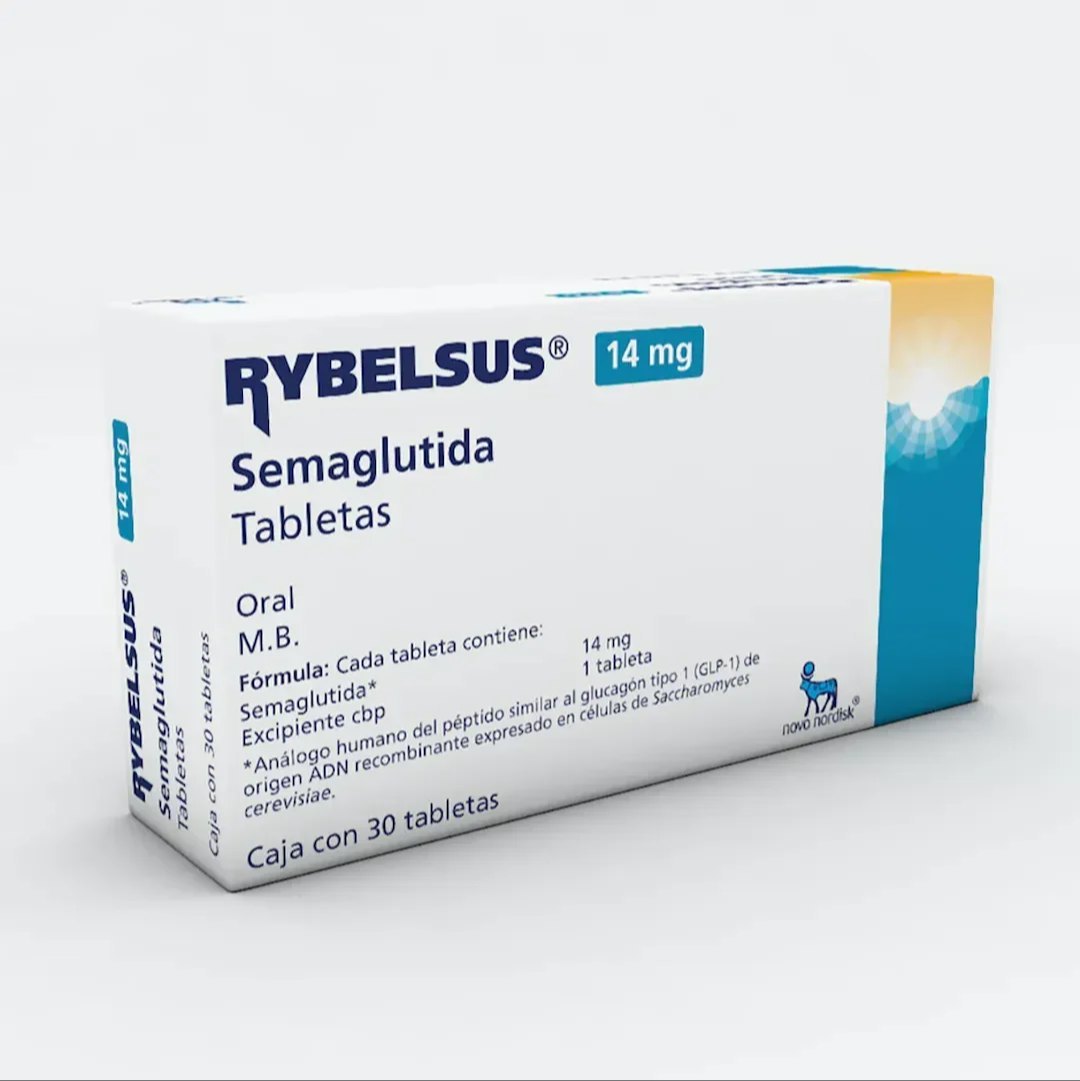
Rybelsus 14mg
$210.00 – $360.00Price range: $210.00 through $360.00
Rybelsus® 14mg is the maximum maintenance dose of the oral GLP-1 receptor agonist for adults with type 2 diabetes. This once-daily tablet provides optimal glycemic control following gradual dose escalation, offering the highest available efficacy in oral GLP-1 therapy as part of a comprehensive diabetes management plan.
Maximum Efficacy in Oral GLP-1 Therapy
Rybelsus® 14mg tablets represent the pinnacle of oral GLP-1 receptor agonist therapy for type 2 diabetes, delivering the highest available dose of oral semaglutide for patients requiring maximal pharmacological support. Reserved for individuals who have not achieved sufficient glycemic control at the 7mg dose despite proper administration and adequate treatment duration, the 14mg strength provides robust GLP-1 receptor activation through convenient once-daily oral administration. This maximum dose amplifies all therapeutic mechanisms—including enhanced glucose-dependent insulin secretion, substantial glucagon suppression, and significant slowing of gastric emptying—while maintaining the unique advantage of non-injectable delivery. The 14mg dose offers patients and providers an oral option with efficacy approaching that of injectable GLP-1 agonists, potentially serving as a comprehensive monotherapy or cornerstone of combination treatment regimens.
Optimal Glycemic Control with Oral Administration Discipline
At the 14mg daily strength, Rybelsus® delivers maximum GLP-1 receptor activation available in oral formulation, translating to clinically significant improvements in glycemic parameters. Clinical trial data demonstrates that the 14mg dose typically achieves A1C reductions of approximately 1.2-1.5% from baseline when used as monotherapy, with many patients reaching their personalized glycemic targets. The continued requirement for strict administration protocol—taking the tablet on an empty stomach with no more than 4 ounces of plain water at least 30 minutes before the first food, beverage, or other oral medications—remains absolutely critical at this dose level to ensure optimal absorption and consistent therapeutic effect. Patients who successfully reach and maintain the 14mg dose have typically established reliable administration routines that support long-term treatment adherence and glycemic stability.
Key Features of Rybelsus® 14mg:
-
Maximum Oral Dose: Provides the highest available GLP-1 receptor activation in oral formulation for patients requiring optimal glycemic control.
-
Proven Efficacy: Demonstrated significant A1C reductions in clinical trials, offering an oral alternative with efficacy approaching injectable GLP-1 agonists.
-
Non-Injectable Convenience: Maintains the unique advantage of oral administration while delivering substantial therapeutic benefits.
-
Comprehensive Mechanism: Addresses multiple pathophysiological aspects of type 2 diabetes through enhanced insulin secretion, glucagon regulation, and gastric motility effects.
-
Long-Term Maintenance Option: Serves as a sustainable oral treatment for patients who achieve glycemic control at this dose level.
Clinical Protocol & Administration Guidelines:
-
Dosage Schedule: One 14mg tablet daily, following at least 30 days at 7mg. Take on an empty stomach with no more than 4 ounces of plain water.
-
Critical Administration Instructions:
-
Take at least 30 minutes BEFORE the first food, beverage, or other oral medications of the day.
-
Swallow tablet whole—do not split, crush, or chew.
-
Wait at least 30 minutes after taking Rybelsus® before eating, drinking (except small sips of water), or taking other oral medications.
-
-
Treatment Evaluation: This dose is intended for long-term maintenance. Patients should be periodically reassessed for continued efficacy, tolerability, and need.
-
Common Side Effects: Nausea, abdominal pain, diarrhea, decreased appetite, vomiting, and constipation. Gastrointestinal effects may temporarily increase with escalation to 14mg but typically diminish over time.
-
Important Safety Information:
-
Boxed Warning: Risk of thyroid C-cell tumors. Contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
-
Pancreatitis: Discontinue if pancreatitis is suspected. Do not restart if pancreatitis is confirmed.
-
Hypoglycemia: Risk increases when used with insulin or insulin secretagogues; may require dose adjustment of these concomitant medications.
-
Diabetic Retinopathy: Monitor patients with a history of diabetic retinopathy.
-
Acute Kidney Injury: Monitor renal function in patients reporting severe gastrointestinal reactions.
-
-
Medical Supervision: Requires ongoing monitoring of glycemic control, potential side effects, and continued adherence to administration instructions.
| QUANTITY | 30, 60, 90 |
|---|
Be the first to review “Rybelsus 14mg” Cancel reply
Related products
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes










Reviews
There are no reviews yet.