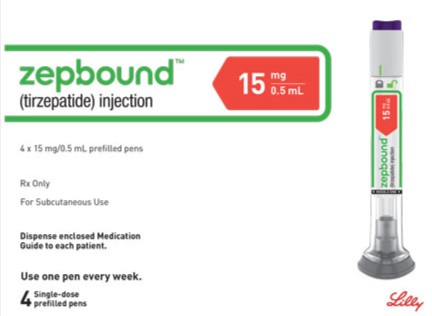
Zepbound 15mg
$360.00
Zepbound™ 15mg is the maximum maintenance dose for chronic weight management in adults with obesity or overweight with weight-related conditions. This once-weekly dual GIP and GLP-1 receptor agonist injection provides optimal appetite regulation and substantial weight reduction following complete dose escalation under ongoing medical supervision as part of a comprehensive weight management program.
Maximum Dual-Incretin Activation for Optimal Weight Management
The Zepbound™ 15mg dose represents the pinnacle of tirzepatide therapy for chronic weight management, delivering the highest available activation of both GIP and GLP-1 receptors for patients who require the most intensive pharmacological support. As the maximum approved dosage in the Zepbound™ range, this weekly injection provides robust, synergistic engagement of dual incretin pathways, offering patients with substantial weight management challenges the greatest opportunity to achieve their treatment goals. Reserved for individuals who have not achieved sufficient weight reduction at lower doses despite adequate titration, the 15mg strength represents a powerful intervention within a comprehensive weight management program that includes reduced-calorie diet, increased physical activity, and behavioral support.
Optimal Synergistic Effects for Transformative Weight Management Outcomes
At the 15mg weekly strength, Zepbound™ achieves maximal therapeutic activation of both incretin pathways, producing amplified effects across all mechanisms of action relevant to weight management. Clinical trial data from the SURMOUNT program demonstrates this dose typically produces the greatest weight loss among all Zepbound™ doses, with patients achieving average reductions of 15-20% of body weight over 72 weeks when combined with lifestyle intervention. The maximal GIP activity contributes to optimal metabolic improvements and may promote favorable body composition changes, while the peak GLP-1 effects provide superior appetite regulation, strong satiety signaling, and significant reduction in both hunger and food cravings. For qualified patients, the 15mg dose offers the potential for transformative weight reduction, frequently enabling achievement of substantial weight loss that can meaningfully impact weight-related health risks and quality of life.
Key Features of the 15mg Maximum Dose:
-
Maximum Dual-Agonist Efficacy: Provides the highest available activation of both GIP and GLP-1 receptors, delivering optimal appetite regulation and weight reduction benefits.
-
Superior Clinical Outcomes: Achieves the greatest weight loss in the Zepbound™ range, as demonstrated in comprehensive clinical trials.
-
Comprehensive Metabolic Impact: Addresses multiple physiological factors involved in weight management, including appetite regulation, metabolic function, and body composition.
-
Advanced Treatment Option: Reserved for patients with inadequate response to lower doses, offering potent therapeutic support within the dual-incretin agonist class.
-
Maintenance Maximization: Allows for full utilization of pharmacological support before considering additional weight management strategies or interventions.
Clinical Protocol & Long-Term Management:
-
Dosage Progression Protocol: Initiated only after patients have completed adequate trials at lower doses (typically at least 4 weeks each at 12.5mg and preceding doses) with documented inadequate response. Administered as one subcutaneous injection of 15mg once weekly.
-
Treatment Evaluation: Patients should be comprehensively assessed after 4-8 weeks at 15mg. This is considered a long-term maintenance dose for most patients, though periodic reevaluation of continued need and tolerability is essential.
-
Administration: Uses a dedicated Zepbound™ single-use pre-filled pen specifically calibrated for 15mg dosing. Inject subcutaneously into abdomen, thigh, or upper arm with weekly site rotation.
-
Side Effect Vigilance: Gastrointestinal effects (particularly nausea, vomiting, diarrhea, and decreased appetite) are most common and potentially most pronounced at this maximum dose. These typically diminish over 4-8 weeks but require proactive management and close patient communication.
-
Comprehensive Monitoring Required: Regular assessment of weight maintenance, nutritional status, potential side effects, and overall health is essential. Monitor for signs of gallbladder disease, pancreatitis, or other medication-related concerns.
-
Mental Health Monitoring: Monitor closely for depression or suicidal thoughts, especially during periods of significant weight change. Ensure immediate access to mental health support resources.
-
Hydration & Nutritional Management: Patients require specific guidance on adequate fluid intake, electrolyte balance, and nutrient-dense meal planning to mitigate side effects and prevent nutritional deficiencies.
-
Absolute Contraindications: Patients with personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Not recommended in patients with severe renal impairment or severe gastrointestinal disease.
-
Risk-Benefit Consideration: Requires careful evaluation of potential benefits versus risks, particularly for patients with history of pancreatitis, gallbladder disease, or significant gastrointestinal disorders.
Be the first to review “Zepbound 15mg” Cancel reply
Related products
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes
Weight loss & T2 Diabetes









Reviews
There are no reviews yet.